Abobotulinumtoxina
Dysport (abobotulinumtoxina) is an unknown pharmaceutical. Abobotulinumtoxina was first approved as Dysport on 2009-04-29. It is used to treat blepharospasm, dystonia, fissure in ano, genetic skin diseases, and hyperhidrosis amongst others in the USA.
Download report
Favorite
Commercial
Therapeutic Areas
Trade Name
FDA
EMA
Dysport
Drug Products
FDA
EMA
Reference product - 351(a)
Reference product - 351(a)
Interchangeable product - 351(k)
Interchangeable product - 351(k)
Biosimilar product - 351(k)
Biosimilar product - 351(k)
Abobotulinumtoxina
Tradename | Proper name | Company | Number | Date | Products |
|---|---|---|---|---|---|
| Dysport | abobotulinumtoxinA | Ipsen Biopharm Limited | N-125274 RX | 2009-04-29 | 2 products |
Labels
FDA
EMA
Brand Name | Status | Last Update |
|---|---|---|
| dysport | Biologic Licensing Application | 2023-01-31 |
Indications
FDA
EMA
Indication | Ontology | MeSH | ICD-10 |
|---|---|---|---|
| blepharospasm | — | D001764 | G24.5 |
| dystonia | HP_0001332 | D004421 | G24 |
| fissure in ano | HP_0012390 | D005401 | K60.2 |
| genetic skin diseases | — | D012873 | — |
| hyperhidrosis | HP_0000975 | D006945 | — |
| muscle rigidity | HP_0002063 | D009127 | — |
| strabismus | HP_0000486 | D013285 | H50.2 |
Agency Specific
FDA
EMA
Expiration | Code | ||
|---|---|---|---|
abobotulinumtoxina, Dysport, Ipsen Biopharm Limited | |||
| 2023-07-29 | Orphan excl. | ||
Patent Expiration
No data
ATC Codes
No data
HCPCS
Code | Description |
|---|---|
| J0586 | Injection, abobotulinumtoxina, 5 units |
Clinical
Clinical Trials
89 clinical trials
View more details

Mock data
Subscribe for the real data
Subscribe for the real data
Indications Phases 4
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Cystic fibrosis | D003550 | EFO_0000390 | E84 | 1 | 1 | — | 1 | 4 | 7 |
| Anesthesia | D000758 | — | 2 | 2 | 1 | 1 | 6 | ||
| Obesity | D009765 | EFO_0001073 | E66.9 | 1 | — | — | 1 | — | 2 |
| Postoperative nausea and vomiting | D020250 | EFO_0004888 | — | — | — | 1 | — | 1 |
Indications Phases 3
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Healthy volunteers/patients | — | 1 | — | 1 | — | 2 | 4 | ||
| Coronary artery disease | D003324 | I25.1 | — | 2 | 1 | — | — | 3 | |
| Coronary artery bypass | D001026 | EFO_0003776 | — | — | 2 | — | — | 2 | |
| Post-cardiac arrest syndrome | D000080942 | — | — | 1 | — | — | 1 | ||
| Coronary disease | D003327 | — | — | 1 | — | — | 1 | ||
| Panic disorder | D016584 | EFO_0004262 | F41.0 | — | 1 | 1 | — | — | 1 |
| Post-traumatic stress disorders | D013313 | EFO_0001358 | F43.1 | — | 1 | 1 | — | — | 1 |
| Pulmonary embolism | D011655 | EFO_0003827 | I26 | — | — | 1 | — | — | 1 |
| Postoperative cognitive complications | D000079690 | — | — | 1 | — | — | 1 | ||
| Diabetic retinopathy | D003930 | EFO_0003770 | — | — | 1 | — | — | 1 |
Show 2 more
Indications Phases 2
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Chronic obstructive pulmonary disease | D029424 | EFO_0000341 | J44.9 | 4 | 2 | — | — | 1 | 6 |
| Idiopathic pulmonary fibrosis | D054990 | J84.112 | 1 | 2 | — | — | — | 3 | |
| Non-small-cell lung carcinoma | D002289 | 1 | 1 | — | — | 1 | 3 | ||
| Brain hypoxia-ischemia | D020925 | EFO_1000846 | P91.6 | 1 | 2 | — | — | 1 | 3 |
| Respiration disorders | D012120 | J00-J99 | 2 | 2 | — | — | — | 2 | |
| Covid-19 | D000086382 | U07.1 | — | 1 | — | — | 1 | 2 | |
| Vaping | D000072137 | 1 | 1 | — | — | 1 | 2 | ||
| Bronchiolitis obliterans | D001989 | EFO_0007183 | 1 | 1 | — | — | 1 | 2 | |
| Colorectal neoplasms | D015179 | — | 1 | — | — | — | 1 | ||
| Delirium | D003693 | R41.0 | — | 1 | — | — | — | 1 |
Show 10 more
Indications Phases 1
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Lung diseases | D008171 | EFO_0003818 | J98.4 | 1 | — | — | — | 1 | 2 |
| Bipolar disorder | D001714 | EFO_0000289 | F30.9 | 1 | — | — | — | — | 1 |
| Major depressive disorder | D003865 | EFO_0003761 | F22 | 1 | — | — | — | — | 1 |
| Alzheimer disease | D000544 | EFO_0000249 | F03 | 1 | — | — | — | — | 1 |
Indications Without Phase
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Premature birth | D047928 | EFO_0003917 | O60 | — | — | — | — | 1 | 1 |
| Bronchopulmonary dysplasia | D001997 | P27.8 | — | — | — | — | 1 | 1 | |
| Emphysema | D004646 | EFO_0000464 | J43 | — | — | — | — | 1 | 1 |
| Cholelithiasis | D002769 | EFO_0004799 | K80 | — | — | — | — | 1 | 1 |
| Cholecystitis | D002764 | HP_0001082 | K81 | — | — | — | — | 1 | 1 |
| Brain diseases | D001927 | HP_0001298 | G93.40 | — | — | — | — | 1 | 1 |
| Marijuana smoking | D008385 | — | — | — | — | 1 | 1 | ||
| Marijuana use | D000074609 | — | — | — | — | 1 | 1 | ||
| Lung injury | D055370 | S27.30 | — | — | — | — | 1 | 1 | |
| Infections | D007239 | EFO_0000544 | — | — | — | — | 1 | 1 |
Show 9 more
Epidemiology
Epidemiological information for investigational and approved indications
View more details
Drug
General
| Drug common name | ABOBOTULINUMTOXINA |
| INN | — |
| Description | Botulinum toxin, or botulinum neurotoxin (BoNT), is a neurotoxic protein produced by the bacterium Clostridium botulinum and related species. It is also known as Botulin, the density of which is predicted to be 1.5±0.1 g/cm3.
It prevents the release of the neurotransmitter acetylcholine from axon endings at the neuromuscular junction, thus causing flaccid paralysis. The toxin causes the disease botulism. The toxin is also used commercially for medical and cosmetic purposes.
|
| Classification | Neurotoxin |
| Drug class | — |
| Image (chem structure or protein) | 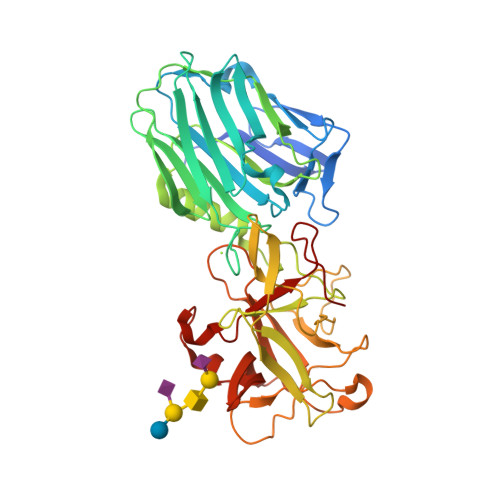 |
| Structure (InChI/SMILES or Protein Sequence) | >2VU9:A|BOTULINUM NEUROTOXIN A HEAVY CHAIN
MGSSHHHHHHSSGLVPRGSHMDTSILNLRYESNHLIDLSRYASKINIGSKVNFDPIDKNQIQLFNLESSKIEVILKNAIV
YNSMYENFSTSFWIRIPKYFNSISLNNEYTIINCMENNSGWKVSLNYGEIIWTLQDTQEIKQRVVFKYSQMINISDYINR
WIFVTITNNRLNNSKIYINGRLIDQKPISNLGNIHASNNIMFKLDGCRDTHRYIWIKYFNLFDKELNEKEIKDLYDNQSN
SGILKDFWGDYLQYDKPYYMLNLYDPNKYVDVNNVGIRGYMYLKGPRGSVMTTNIYLNSSLYRGTKFIIKKYASGNKDNI
VRNNDRVYINVVVKNKEYRLATNASQAGVEKILSALEIPDVGNLSQVVVMKSKNDQGITNKCKMNLQDNNGNDIGFIGFH
QFNNIAKLVASNWYNRQIERSSRTLGCSWEFIPVDDGWGERPLQ |
Identifiers
| PDB | 2G7P, 2VU9, 2VUA, 3BTA, 3DDA, 3DDB, 3DS9, 3DSE, 3FUO, 3K3Q, 3QIX, 3QIY, 3QIZ, 3QJ0, 3QW5, 3QW6, 3QW7, 3QW8, 3ZUR, 3ZUS, 4EJ5, 4EL4, 4ELC, 4HEV, 4IQP, 4JRA, 4KS6, 4KTX, 4KUF, 4ZJX, 5JLV, 5JMC, 5L21, 5MK6, 5MK7, 5MOY, 5TPB, 5TPC, 5V8P, 5V8R, 5V8U, 5VGV, 5VGX, 6DKK, 6ES1, 6MHJ, 6XCB, 6XCC, 6XCD, 6XCE, 6XCF, 7N18 |
| CAS-ID | — |
| RxCUI | — |
| ChEMBL ID | CHEMBL2108570 |
| ChEBI ID | — |
| PubChem CID | — |
| DrugBank | DB00083 |
| UNII ID | — |
Target
Agency Approved
No data
Alternate
No data
Variants
Clinical Variant
No data
Financial
No data
Trends
PubMed Central
Top Terms for Disease or Syndrome:

Mock data
Subscribe for the real data
Subscribe for the real data
Additional graphs summarizing 1,539 documents
View more details
Safety
Black-box Warning
Black-box warning for: Dysport
Adverse Events
Top Adverse Reactions

Mock data
Subscribe for the real data
Subscribe for the real data
7 adverse events reported
View more details
Premium feature
Learn more about premium features at pharmakb.com
Learn more
