Campath , Mabcampath(alemtuzumab)
Campath, Lemtrada (alemtuzumab) is an antibody pharmaceutical. Alemtuzumab was first approved as Campath on 2001-05-07. It is used to treat b-cell chronic lymphocytic leukemia in the USA. It has been approved in Europe to treat b-cell chronic lymphocytic leukemia and multiple sclerosis. The pharmaceutical is active against CAMPATH-1 antigen.
Download report
Favorite
Commercial
Trade Name
FDA
EMA
Campath, Lemtrada
Drug Products
FDA
EMA
Reference product - 351(a)
Reference product - 351(a)
Interchangeable product - 351(k)
Interchangeable product - 351(k)
Biosimilar product - 351(k)
Biosimilar product - 351(k)
Agency Specific
FDA
EMA
Expiration | Code | ||
|---|---|---|---|
alemtuzumab, Campath , Genzyme Corporation | |||
| 2108-05-07 | Orphan excl. | ||
alemtuzumab, Lemtrada, Genzyme Corporation | |||
| 2108-05-07 | Orphan excl. | ||
Patent Expiration
No data
HCPCS
Code | Description |
|---|---|
| J0202 | Injection, alemtuzumab, 1 mg |
Clinical
Clinical Trials
304 clinical trials
View more details

Mock data
Subscribe for the real data
Subscribe for the real data
Indications Phases 4
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Multiple sclerosis | D009103 | EFO_0003885 | G35 | 3 | 2 | 3 | 2 | 8 | 17 |
| Kidney transplantation | D016030 | 3 | 6 | 2 | 5 | 3 | 16 | ||
| Relapsing-remitting multiple sclerosis | D020529 | EFO_0003929 | — | 2 | 4 | 3 | — | 9 | |
| Kidney diseases | D007674 | EFO_0003086 | N08 | 2 | 3 | — | 1 | — | 4 |
| Renal insufficiency | D051437 | HP_0000083 | N19 | 2 | 2 | — | 2 | — | 4 |
| Chronic kidney failure | D007676 | EFO_0003884 | N18.6 | 1 | 2 | — | 1 | — | 3 |
| Graft rejection | D006084 | — | 1 | — | 1 | 1 | 3 | ||
| Vasculitis | D014657 | EFO_0006803 | M31 | 1 | — | — | 1 | — | 2 |
| Chronic inflammatory demyelinating polyradiculoneuropathy | D020277 | EFO_1000868 | G61.81 | — | — | — | 1 | — | 1 |
| Granulomatosis with polyangiitis | D014890 | EFO_0005297 | M31.3 | — | — | — | 1 | — | 1 |
Show 2 more
Indications Phases 3
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Leukemia | D007938 | C95 | 10 | 38 | 2 | — | 3 | 44 | |
| B-cell chronic lymphocytic leukemia | D015451 | C91.1 | 7 | 28 | 2 | — | 5 | 38 | |
| Sickle cell anemia | D000755 | EFO_0000697 | D57 | 11 | 20 | 2 | — | 1 | 26 |
| Myelodysplastic syndromes | D009190 | D46 | 6 | 20 | 2 | — | — | 22 | |
| Aplastic anemia | D000741 | HP_0001915 | D61.9 | 2 | 10 | 1 | — | 2 | 13 |
| Myeloid leukemia acute | D015470 | C92.0 | 3 | 7 | 1 | — | — | 9 | |
| T-cell lymphoma peripheral | D016411 | C84.9 | 2 | 7 | 1 | — | — | 9 | |
| Thalassemia | D013789 | EFO_1001996 | D56 | — | 6 | 1 | — | 1 | 7 |
| Extranodal nk-t-cell lymphoma | D054391 | C86.0 | — | 3 | 1 | — | — | 4 | |
| Diamond-blackfan anemia | D029503 | D61.01 | 1 | 3 | 1 | — | — | 3 |
Show 6 more
Indications Phases 2
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Lymphoma | D008223 | C85.9 | 9 | 24 | — | — | 2 | 30 | |
| Precursor cell lymphoblastic leukemia-lymphoma | D054198 | C91.0 | 6 | 14 | — | — | 1 | 16 | |
| Non-hodgkin lymphoma | D008228 | C85.9 | 8 | 9 | — | — | 2 | 15 | |
| Hodgkin disease | D006689 | C81 | 3 | 11 | — | — | 1 | 12 | |
| Graft vs host disease | D006086 | D89.81 | 3 | 7 | — | — | 1 | 10 | |
| Myeloproliferative disorders | D009196 | D47.1 | 3 | 6 | — | — | 1 | 9 | |
| Multiple myeloma | D009101 | C90.0 | 4 | 9 | — | — | — | 9 | |
| Bcr-abl positive chronic myelogenous leukemia | D015464 | EFO_0000340 | 3 | 8 | — | — | — | 8 | |
| Lymphoproliferative disorders | D008232 | Orphanet_2442 | D47.9 | 2 | 7 | — | — | 1 | 8 |
| Hematologic neoplasms | D019337 | 4 | 5 | — | — | 1 | 7 |
Show 104 more
Indications Phases 1
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Melanoma | D008545 | 1 | — | — | — | — | 1 | ||
| Germ cell and embryonal neoplasms | D009373 | 1 | — | — | — | — | 1 | ||
| Gastrointestinal stromal tumors | D046152 | EFO_0000505 | C49.A | 1 | — | — | — | — | 1 |
| Rhabdoid tumor | D018335 | 1 | — | — | — | — | 1 | ||
| Ewing sarcoma | D012512 | EFO_0000173 | 1 | — | — | — | — | 1 | |
| Rhabdomyosarcoma | D012208 | 1 | — | — | — | — | 1 | ||
| Large cell carcinoma | D018287 | 1 | — | — | — | — | 1 | ||
| Wilms tumor | D009396 | 1 | — | — | — | — | 1 | ||
| Neuroepithelial neoplasms | D018302 | 1 | — | — | — | — | 1 | ||
| Rheumatoid arthritis | D001172 | EFO_0000685 | M06.9 | 1 | — | — | — | — | 1 |
Show 5 more
Indications Without Phase
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Lysosomal storage diseases | D016464 | — | — | — | — | 1 | 1 | ||
| Hematopoietic stem cell transplantation | D018380 | — | — | — | — | 1 | 1 | ||
| Hemoglobin sc disease | D006450 | EFO_1001797 | D57.2 | — | — | — | — | 1 | 1 |
| Kidney neoplasms | D007680 | EFO_0003865 | C64 | — | — | — | — | 1 | 1 |
| B-cell lymphoma | D016393 | — | — | — | — | 1 | 1 |
Epidemiology
Epidemiological information for investigational and approved indications
View more details
Drug
General
| Drug common name | ALEMTUZUMAB |
| INN | alemtuzumab |
| Description | Immunoglobulin G 1 (human-rat monodonal CAMPATH-1H 71-chain anti-human antigen CD52), disulfide with human-rat monoclonal CAMPATH-1H light chain, dimer |
| Classification | Antibody |
| Drug class | monoclonal antibodies |
| Image (chem structure or protein) | 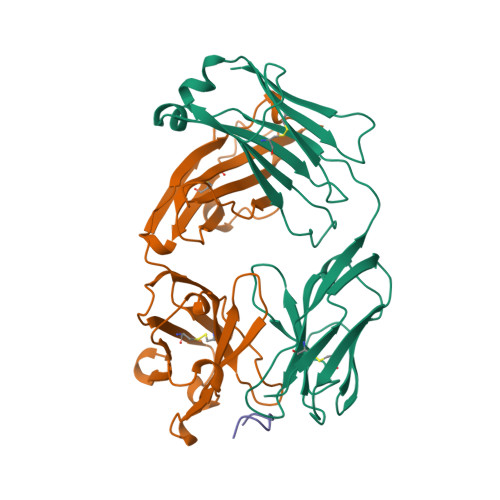 |
| Structure (InChI/SMILES or Protein Sequence) | >1CE1:H|PROTEIN (CAMPATH-1H:HEAVY CHAIN)
QVQLQESGPGLVRPSQTLSLTCTVSGFTFTDFYMNWVRQPPGRGLEWIGFIRDKAKGYTTEYNPSVKGRVTMLVDTSKNQ
FSLRLSSVTAADTAVYYCAREGHTAAPFDYWGQGSLVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTV
SWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKVE
>1CE1:L|PROTEIN (CAMPATH-1H:LIGHT CHAIN)
DIQMTQSPSSLSASVGDRVTITCKASQNIDKYLNWYQQKPGKAPKLLIYNTNNLQTGVPSRFSGSGSGTDFTFTISSLQP
EDIATYYCLQHISRPRTFGQGTKVEIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQ
ESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNR |
Identifiers
| PDB | 1BEY, 1BFO, 1CE1, 6OBD |
| CAS-ID | 216503-57-0 |
| RxCUI | 117055 |
| ChEMBL ID | CHEMBL1201587 |
| ChEBI ID | — |
| PubChem CID | — |
| DrugBank | DB00087 |
| UNII ID | 3A189DH42V (ChemIDplus, GSRS) |
Target
Agency Approved
CD52
CD52
Organism
Homo sapiens
Gene name
CD52
Gene synonyms
CDW52, HE5
NCBI Gene ID
Protein name
CAMPATH-1 antigen
Protein synonyms
Cambridge pathology 1 antigen, CD52, CD52 antigen (CAMPATH-1 antigen), CDw52, CDW52 antigen (CAMPATH-1 antigen), Epididymal secretory protein E5, epididymis secretory sperm binding protein Li 171mP, He5, HEL-S-171mP, Human epididymis-specific protein 5
Uniprot ID
Mouse ortholog
Cd52 (23833)
CAMPATH-1 antigen (Q64389)
Alternate
No data
Variants
Clinical Variant
No data
Financial
No data
Trends
PubMed Central
Top Terms for Disease or Syndrome:

Mock data
Subscribe for the real data
Subscribe for the real data
Additional graphs summarizing 11,703 documents
View more details
Safety
Black-box Warning
Black-box warning for: Campath, Lemtrada
Adverse Events
Top Adverse Reactions

Mock data
Subscribe for the real data
Subscribe for the real data
3,660 adverse events reported
View more details
Premium feature
Learn more about premium features at pharmakb.com
Learn more
