Rybrevant(amivantamab)
Rybrevant (amivantamab) is an antibody pharmaceutical. Amivantamab was first approved as Rybrevant on 2021-05-21. It is used to treat non-small-cell lung carcinoma in the USA. It has been approved in Europe to treat non-small-cell lung carcinoma. The pharmaceutical is active against epidermal growth factor receptor and hepatocyte growth factor receptor.
Download report
Favorite
Commercial
Trade Name
FDA
EMA
Rybrevant
Drug Products
FDA
EMA
Reference product - 351(a)
Reference product - 351(a)
Interchangeable product - 351(k)
Interchangeable product - 351(k)
Biosimilar product - 351(k)
Biosimilar product - 351(k)
Amivantamab
Tradename | Proper name | Company | Number | Date | Products |
|---|---|---|---|---|---|
| Rybrevant | amivantamab-vmjw | Johnson & Johnson | N-761210 RX | 2021-05-21 | 1 products |
Labels
FDA
EMA
Brand Name | Status | Last Update |
|---|---|---|
| rybrevant | Biologic Licensing Application | 2022-01-24 |
Indications
FDA
EMA
Indication | Ontology | MeSH | ICD-10 |
|---|---|---|---|
| non-small-cell lung carcinoma | — | D002289 | — |
Agency Specific
FDA
EMA
No data
Patent Expiration
No data
HCPCS
Code | Description |
|---|---|
| J9061 | Injection, amivantamab-vmjw, 2 mg |
Clinical
Clinical Trials
23 clinical trials
View more details

Mock data
Subscribe for the real data
Subscribe for the real data
Indications Phases 4
No data
Indications Phases 3
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Non-small-cell lung carcinoma | D002289 | 4 | 6 | 4 | — | 1 | 14 |
Indications Phases 2
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Colorectal neoplasms | D015179 | 2 | 1 | — | — | — | 2 | ||
| Neoplasms | D009369 | C80 | — | 1 | — | — | — | 1 | |
| Stomach neoplasms | D013274 | EFO_0003897 | C16 | — | 1 | — | — | — | 1 |
| Esophageal neoplasms | D004938 | C15 | — | 1 | — | — | — | 1 | |
| Salivary gland neoplasms | D012468 | EFO_0003826 | D11 | — | 1 | — | — | — | 1 |
| Liver neoplasms | D008113 | EFO_1001513 | C22.0 | — | 1 | — | — | — | 1 |
Indications Phases 1
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Breast neoplasms | D001943 | EFO_0003869 | C50 | 1 | — | — | — | — | 1 |
| Ovarian neoplasms | D010051 | EFO_0003893 | C56 | 1 | — | — | — | — | 1 |
| Pancreatic neoplasms | D010190 | EFO_0003860 | C25 | 1 | — | — | — | — | 1 |
| Head and neck neoplasms | D006258 | 1 | — | — | — | — | 1 |
Indications Without Phase
No data
Epidemiology
Epidemiological information for investigational and approved indications
View more details
Drug
General
| Drug common name | AMIVANTAMAB |
| INN | amivantamab |
| Description | Amivantamab, sold under the brand name Rybrevant, is a bispecific monoclonal antibody used to treat non-small cell lung cancer. Amivantamab is a bispecific epidermal growth factor (EGF) receptor-directed and mesenchymal–epithelial transition (MET) receptor-directed antibody. It is the first treatment for adults with non-small cell lung cancer whose tumors have specific types of genetic mutations: epidermal growth factor receptor (EGFR) exon 20 insertion mutations.
|
| Classification | Antibody |
| Drug class | monoclonal antibodies |
| Image (chem structure or protein) | 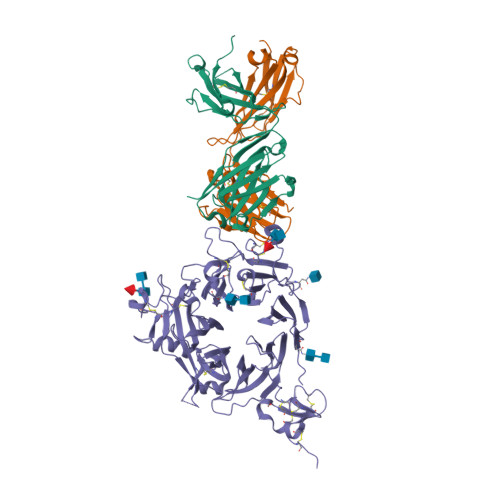 |
| Structure (InChI/SMILES or Protein Sequence) | >6WVZ:H|Heavy Chain of anti-MET Fab of amivantamab
QVQLVQSGAEVKKPGASVKVSCETSGYTFTSYGISWVRQAPGHGLEWMGWISAYNGYTNYAQKLQGRVTMTTDTSTSTAY
MELRSLRSDDTAVYYCARDLRGTNYFDYWGQGTLVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSW
NSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSCHHHHHH
>6WVZ:L|Light Chain of anti-MET Fab of amivantamab
DIQMTQSPSSVSASVGDRVTITCRASQGISNWLAWFQHKPGKAPKLLIYAASSLLSGVPSRFSGSGSGTDFTLTISSLQP
EDFATYYCQQANSFPITFGQGTRLEIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQ
ESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC |
Identifiers
| PDB | 6WVZ |
| CAS-ID | 2171511-58-1 |
| RxCUI | — |
| ChEMBL ID | CHEMBL4297774 |
| ChEBI ID | — |
| PubChem CID | — |
| DrugBank | DB16695 |
| UNII ID | 0JSR7Z0NB6 (ChemIDplus, GSRS) |
Target
Agency Approved
EGFR
EGFR
MET
MET
Organism
Homo sapiens
Gene name
EGFR
Gene synonyms
ERBB, ERBB1, HER1
NCBI Gene ID
Protein name
epidermal growth factor receptor
Protein synonyms
avian erythroblastic leukemia viral (v-erb-b) oncogene homolog, cell growth inhibiting protein 40, cell proliferation-inducing protein 61, EGFR vIII, epidermal growth factor receptor tyrosine kinase domain, erb-b2 receptor tyrosine kinase 1, Proto-oncogene c-ErbB-1, Receptor tyrosine-protein kinase erbB-1
Uniprot ID
Mouse ortholog
Egfr (13649)
epidermal growth factor receptor (Q01279)
Alternate
No data
Variants
Clinical Variant
No data
Financial
No data
Trends
PubMed Central
Top Terms for Disease or Syndrome:

Mock data
Subscribe for the real data
Subscribe for the real data
Additional graphs summarizing 399 documents
View more details
Safety
Black-box Warning
No Black-box warning
Adverse Events
Top Adverse Reactions

Mock data
Subscribe for the real data
Subscribe for the real data
59 adverse events reported
View more details
Premium feature
Learn more about premium features at pharmakb.com
Learn more
