Ilaris(canakinumab)
Ilaris (canakinumab) is an antibody pharmaceutical. Canakinumab was first approved as Ilaris on 2009-06-17. It is used to treat cryopyrin-associated periodic syndromes, familial mediterranean fever, juvenile arthritis, and mevalonate kinase deficiency in the USA. It has been approved in Europe to treat cryopyrin-associated periodic syndromes, gouty arthritis, and juvenile arthritis. The pharmaceutical is active against interleukin-1 beta.
Download report
Favorite
COVID-19
Novartis Pharmaceuticals
Top 200 Pharmaceuticals by Retail Sales
Commercial
Therapeutic Areas
Therapeutic Area | MeSH |
|---|---|
| musculoskeletal diseases | D009140 |
| nervous system diseases | D009422 |
| hemic and lymphatic diseases | D006425 |
| hereditary congenital and neonatal diseases and abnormalities | D009358 |
| skin and connective tissue diseases | D017437 |
| nutritional and metabolic diseases | D009750 |
| immune system diseases | D007154 |
| signs and symptoms pathological conditions | D013568 |
Trade Name
FDA
EMA
Ilaris
Drug Products
FDA
EMA
Reference product - 351(a)
Reference product - 351(a)
Interchangeable product - 351(k)
Interchangeable product - 351(k)
Biosimilar product - 351(k)
Biosimilar product - 351(k)
Canakinumab
Tradename | Proper name | Company | Number | Date | Products |
|---|---|---|---|---|---|
| Ilaris | canakinumab | Novartis Pharmaceuticals Corporation | N-125319 RX | 2016-12-22 | 1 products |
Show 1 discontinued
Labels
FDA
EMA
Brand Name | Status | Last Update |
|---|---|---|
| ilaris | Biologic Licensing Application | 2020-09-18 |
Agency Specific
FDA
EMA
Expiration | Code | ||
|---|---|---|---|
canakinumab, Ilaris, Novartis Pharmaceuticals Corporation | |||
| 2027-06-16 | Orphan excl. | ||
Patent Expiration
No data
HCPCS
Code | Description |
|---|---|
| J0638 | Injection, canakinumab, 1 mg |
Clinical
Clinical Trials
102 clinical trials
View more details

Mock data
Subscribe for the real data
Subscribe for the real data
Indications Phases 4
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Rheumatoid arthritis | D001172 | EFO_0000685 | M06.9 | 2 | 6 | — | 1 | — | 7 |
| Psoriatic arthritis | D015535 | EFO_0003778 | L40.5 | — | — | — | 1 | — | 1 |
| Spondylarthritis | D025241 | — | — | — | 1 | — | 1 |
Indications Phases 3
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Juvenile arthritis | D001171 | EFO_0002609 | M08 | 1 | 1 | 7 | — | — | 8 |
| Gouty arthritis | D015210 | — | 2 | 5 | — | — | 7 | ||
| Cryopyrin-associated periodic syndromes | D056587 | M04.2 | — | — | 7 | — | — | 7 | |
| Non-small-cell lung carcinoma | D002289 | 1 | 2 | 3 | — | — | 6 | ||
| Type 2 diabetes mellitus | D003924 | EFO_0001360 | E11 | — | 4 | 2 | — | — | 5 |
| Adult-onset still's disease | D016706 | EFO_0007135 | M06.1 | — | 1 | 1 | — | — | 2 |
| Cytokine release syndrome | D000080424 | D89.83 | — | — | 1 | — | 1 | 2 | |
| Atherosclerosis | D050197 | EFO_0003914 | I25.1 | — | 1 | 1 | — | — | 2 |
| Lung neoplasms | D008175 | C34.90 | — | 1 | 1 | — | — | 2 | |
| Pancreatic neoplasms | D010190 | EFO_0003860 | C25 | — | — | 1 | — | — | 1 |
Show 3 more
Indications Phases 2
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Myelodysplastic syndromes | D009190 | D46 | 2 | 3 | — | — | — | 4 | |
| Schnitzler syndrome | D019873 | EFO_1001165 | — | 3 | — | — | — | 3 | |
| Covid-19 | D000086382 | U07.1 | — | 1 | — | — | 1 | 2 | |
| Type 1 diabetes mellitus | D003922 | EFO_0001359 | E10 | — | 2 | — | — | — | 2 |
| Melanoma | D008545 | — | 1 | — | — | — | 1 | ||
| Familial mediterranean fever | D010505 | M04.1 | — | 1 | — | — | — | 1 | |
| Gout | D006073 | EFO_0004274 | M10 | — | 1 | — | — | — | 1 |
| Glucose intolerance | D018149 | HP_0000833 | R73.03 | — | 1 | — | — | — | 1 |
| Knee osteoarthritis | D020370 | EFO_0004616 | M17 | — | 1 | — | — | — | 1 |
| Alzheimer disease | D000544 | EFO_0000249 | F03 | — | 1 | — | — | — | 1 |
Show 29 more
Indications Phases 1
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Triple negative breast neoplasms | D064726 | 2 | — | — | — | — | 2 | ||
| Pancreatic ductal carcinoma | D021441 | 1 | — | — | — | — | 1 | ||
| Renal cell carcinoma | D002292 | 1 | — | — | — | — | 1 | ||
| Macular degeneration | D008268 | EFO_0001365 | H35.30 | 1 | — | — | — | — | 1 |
| Healthy volunteers/patients | — | 1 | — | — | — | — | 1 | ||
| Fever | D005334 | HP_0001945 | R50.9 | 1 | — | — | — | — | 1 |
Indications Without Phase
No data
Epidemiology
Epidemiological information for investigational and approved indications
View more details
Drug
General
| Drug common name | CANAKINUMAB |
| INN | canakinumab |
| Description | Immunoglobulin G1, anti-[Homo sapiens interleukin 1, beta (IL1B)] human monoclonal ACZ885; gamma1 heavy chain (Homo sapiens VH-IGHG1*03) (221-214’)-disulfide with kappa light chain (Homo sapiens V-KAPPA-IGKC*01); (227-227’’:230-230’’)-bisdisulfide dimer |
| Classification | Antibody |
| Drug class | monoclonal antibodies |
| Image (chem structure or protein) | 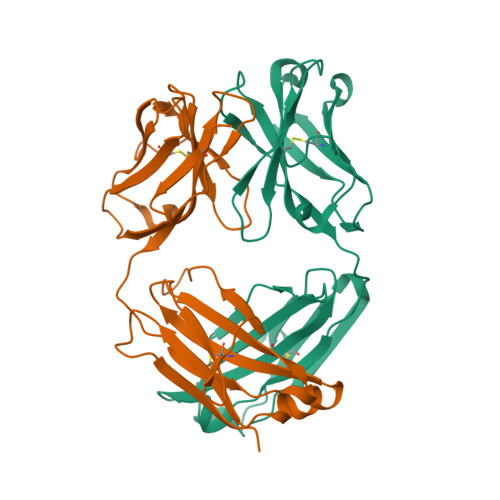 |
| Structure (InChI/SMILES or Protein Sequence) | >4G5Z:H|canakinumab antibody fragment heavy chain
QVQLVESGGGVVQPGRSLRLSCAASGFTFSVYGMNWVRQAPAKGLEWVAIIWYDGDNQYYADSVKGRFTISRDNSKNTLY
LQMNGLRAEDTAVYYCARDLRTGPFDYWGQGTLVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWN
SGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVAP
>4G5Z:L|canakinumab antibody fragment light chain
EIVLTQSPDFQSVTPKEKVTITCRASQSIGSSLHWYQQKPDQSPKLLIKYASQSFSGVPSRFSGSGSGTDFTLTINSLEA
EDAAAYYCHQSSSLPFTFGPGTKVDIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQ
ESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRG |
Identifiers
| PDB | 4G5Z, 4G6J, 5BVJ, 5BVP |
| CAS-ID | 914613-48-2 |
| RxCUI | 853491 |
| ChEMBL ID | CHEMBL1201834 |
| ChEBI ID | — |
| PubChem CID | — |
| DrugBank | DB06168 |
| UNII ID | 37CQ2C7X93 (ChemIDplus, GSRS) |
Target
Alternate
No data
Variants
Clinical Variant
No data
Financial
Ilaris - Novartis
$
€
£
₣

Mock data
Subscribe for the real data
Subscribe for the real data

Mock data
Subscribe for the real data
Subscribe for the real data
Tabular view
Trends
PubMed Central
Top Terms for Disease or Syndrome:

Mock data
Subscribe for the real data
Subscribe for the real data
Additional graphs summarizing 7,397 documents
View more details
Safety
Black-box Warning
No Black-box warning
Adverse Events
Top Adverse Reactions

Mock data
Subscribe for the real data
Subscribe for the real data
2,849 adverse events reported
View more details
Premium feature
Learn more about premium features at pharmakb.com
Learn more
