Opdivo(nivolumab)
Opdivo, Opdualag (nivolumab) is an antibody pharmaceutical. Nivolumab was first approved as Opdivo on 2014-12-22. It is used to treat kidney neoplasms, lymphoma, melanoma, non-small-cell lung carcinoma, and squamous cell carcinoma amongst others in the USA. It has been approved in Europe to treat colorectal neoplasms, hodgkin disease, melanoma, mesothelioma, and non-small-cell lung carcinoma amongst others. The pharmaceutical is active against programmed cell death protein 1.
Download report
Favorite
Drugs Approved for Childhood Cancers
BMS
Top 200 Pharmaceuticals by Retail Sales
Commercial
Trade Name
FDA
EMA
Opdivo
CombinationsOpdualag
Drug Products
FDA
EMA
Reference product - 351(a)
Reference product - 351(a)
Interchangeable product - 351(k)
Interchangeable product - 351(k)
Biosimilar product - 351(k)
Biosimilar product - 351(k)
Agency Specific
FDA
EMA
Expiration | Code | ||
|---|---|---|---|
nivolumab, Opdivo, Bristol-Myers Squibb Company | |||
| 2030-02-15 | Orphan excl. | ||
nivolumab / relatlimab, Opdualag, Bristol-Myers Squibb Company | |||
| 2029-03-18 | Orphan excl. | ||
Patent Expiration
No data
HCPCS
Code | Description |
|---|---|
| J9299 | Injection, nivolumab, 1 mg |
Clinical
Clinical Trials
1411 clinical trials
View more details

Mock data
Subscribe for the real data
Subscribe for the real data
Indications Phases 4
Indications Phases 3
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Neoplasms | D009369 | C80 | 135 | 109 | 4 | — | 2 | 183 | |
| Colorectal neoplasms | D015179 | 36 | 48 | 4 | — | 1 | 71 | ||
| Squamous cell carcinoma of head and neck | D000077195 | 32 | 41 | 4 | — | 4 | 68 | ||
| Hodgkin disease | D006689 | C81 | 20 | 31 | 2 | — | — | 42 | |
| Breast neoplasms | D001943 | EFO_0003869 | C50 | 16 | 20 | 1 | — | 1 | 35 |
| Stomach neoplasms | D013274 | EFO_0003897 | C16 | 14 | 20 | 7 | — | — | 34 |
| Head and neck neoplasms | D006258 | 14 | 20 | 4 | — | 1 | 32 | ||
| Prostatic neoplasms | D011471 | C61 | 10 | 25 | 1 | — | — | 30 | |
| Urinary bladder neoplasms | D001749 | C67 | 10 | 19 | 3 | — | — | 30 | |
| Urologic neoplasms | D014571 | C64-C68 | 17 | 18 | 1 | — | — | 28 |
Show 25 more
Indications Phases 2
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Pancreatic neoplasms | D010190 | EFO_0003860 | C25 | 32 | 33 | — | — | — | 53 |
| Ovarian neoplasms | D010051 | EFO_0003893 | C56 | 21 | 19 | — | — | — | 31 |
| Small cell lung carcinoma | D055752 | 13 | 19 | — | — | — | 24 | ||
| Triple negative breast neoplasms | D064726 | 16 | 10 | — | — | — | 19 | ||
| Castration-resistant prostatic neoplasms | D064129 | 9 | 10 | — | — | — | 17 | ||
| Large b-cell lymphoma diffuse | D016403 | C83.3 | 12 | 8 | — | — | — | 16 | |
| Uveal neoplasms | D014604 | EFO_1001230 | 9 | 10 | — | — | — | 16 | |
| Hepatocellular carcinoma | D006528 | C22.0 | 8 | 11 | — | — | — | 15 | |
| Uterine cervical neoplasms | D002583 | 6 | 10 | — | — | 1 | 14 | ||
| Lymphoma | D008223 | C85.9 | 8 | 7 | — | — | — | 13 |
Show 161 more
Indications Phases 1
Indication | MeSH | Ontology | ICD-10 | Ph 1 | Ph 2 | Ph 3 | Ph 4 | Other | Total |
|---|---|---|---|---|---|---|---|---|---|
| Kaposi sarcoma | D012514 | C46 | 4 | — | — | — | — | 4 | |
| Bcr-abl positive chronic myelogenous leukemia | D015464 | EFO_0000340 | 3 | — | — | — | — | 3 | |
| Anemia | D000740 | EFO_0004272 | D64.9 | 2 | — | — | — | — | 2 |
| Psoriasis | D011565 | EFO_0000676 | L40 | 2 | — | — | — | — | 2 |
| Ulcerative colitis | D003093 | EFO_0000729 | K51 | 2 | — | — | — | — | 2 |
| Rheumatoid arthritis | D001172 | EFO_0000685 | M06.9 | 2 | — | — | — | — | 2 |
| Multiple sclerosis | D009103 | EFO_0003885 | G35 | 2 | — | — | — | — | 2 |
| Crohn disease | D003424 | EFO_0000384 | K50 | 2 | — | — | — | — | 2 |
| Autoimmune diseases | D001327 | EFO_0000540 | M30-M36 | 2 | — | — | — | — | 2 |
| Systemic lupus erythematosus | D008180 | EFO_0002690 | M32 | 2 | — | — | — | — | 2 |
Show 27 more
Epidemiology
Epidemiological information for investigational and approved indications
View more details
Drug
General
| Drug common name | NIVOLUMAB |
| INN | nivolumab |
| Description | Nivolumab, sold under the brand name Opdivo, is a medication used to treat a number of types of cancer. This includes melanoma, lung cancer, renal cell carcinoma, Hodgkin lymphoma, head and neck cancer, colon cancer, and liver cancer. It is used by slow injection into a vein.
|
| Classification | Antibody |
| Drug class | monoclonal antibodies |
| Image (chem structure or protein) | 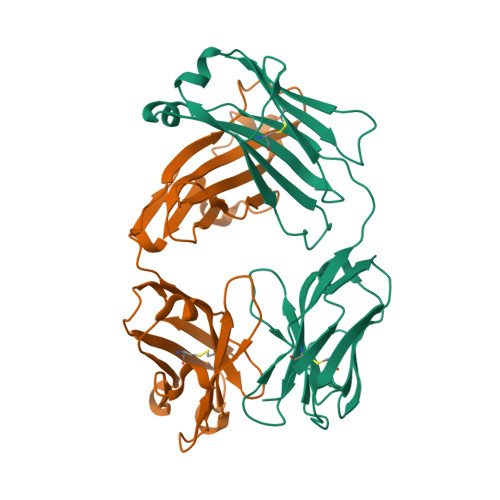 |
| Structure (InChI/SMILES or Protein Sequence) | >5GGQ:H|nivolumab heavy chain
QVQLVESGGGVVQPGRSLRLDCKASGITFSNSGMHWVRQAPGKGLEWVAVIWYDGSKRYYADSVKGRFTISRDNSKNTLF
LQMNSLRAEDTAVYYCATNDDYWGQGTLVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALT
SGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHHHHHH
>5GGQ:L|nivolumab light chain
EIVLTQSPATLSLSPGERATLSCRASQSVSSYLAWYQQKPGQAPRLLIYDASNRATGIPARFSGSGSGTDFTLTISSLEP
EDFAVYYCQQSSNWPRTFGQGTKVEIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQ
ESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC |
Identifiers
| PDB | 5GGQ, 5GGR, 5WT9 |
| CAS-ID | 946414-94-4 |
| RxCUI | 1597876 |
| ChEMBL ID | CHEMBL2108738 |
| ChEBI ID | — |
| PubChem CID | — |
| DrugBank | DB09035 |
| UNII ID | 31YO63LBSN (ChemIDplus, GSRS) |
Target
Agency Approved
PDCD1
PDCD1
Alternate
No data
Variants
Clinical Variant
No data
Financial
Opdivo - Bristol Myers Squibb
$
€
£
₣

Mock data
Subscribe for the real data
Subscribe for the real data

Mock data
Subscribe for the real data
Subscribe for the real data
Opdualag - Bristol Myers Squibb
$
€
£
₣

Mock data
Subscribe for the real data
Subscribe for the real data

Mock data
Subscribe for the real data
Subscribe for the real data
Tabular view
Trends
PubMed Central
Top Terms for Disease or Syndrome:

Mock data
Subscribe for the real data
Subscribe for the real data
Additional graphs summarizing 48,178 documents
View more details
Safety
Black-box Warning
No Black-box warning
Adverse Events
Top Adverse Reactions

Mock data
Subscribe for the real data
Subscribe for the real data
424 adverse events reported
View more details
Premium feature
Learn more about premium features at pharmakb.com
Learn more
